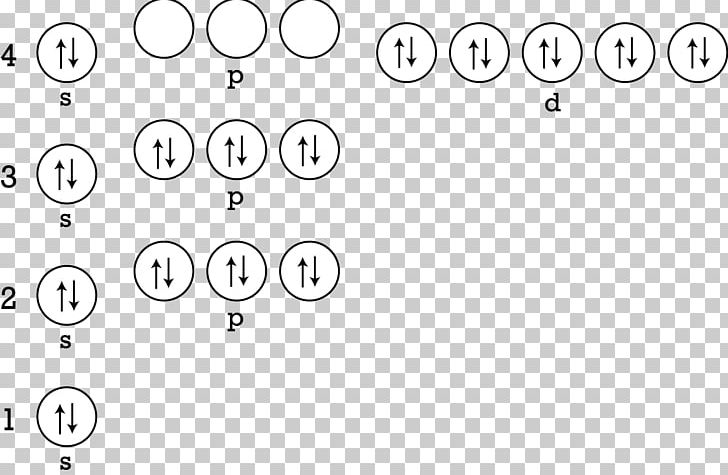
Because the Zn(s) + Cu 2 + (aq) system is higher in energy by 1.10 V than the Cu(s) + Zn 2 + (aq) system, energy is released when electrons are transferred from Zn to Cu 2 + to form Cu and Zn 2 +. In an atomic ion only the number of electrons changes but the number of protons and neutrons does not change. The ionic properties of the elements depend on the exchange of electrons. When a transition metal loses electrons, it tends to lose its s orbital electrons before any of its d orbital electrons. When an atom carries a negative or positive charge by accepting or rejecting electrons, it is called an ion. Since the total number of electrons lost always equals the total number of electrons gained, the compound formed will contain Mg2+ ion(s) for every Cl- ion(s). An atom that accepts an electron to achieve a more stable configuration is assigned an oxidation number of -1. Cl gains electron(s) in the 3p orbital to form the Cl- anion. This desire of atoms to have eight electrons in their outermost shell is known as the octet rule. Much of this potential energy difference is because the valence electrons of metallic Zn are higher in energy than the valence electrons of metallic Cu. When the elements Mg and Cl form an ionic bond, Mg loses electron(s) from the 3s orbital to form the Mg2+ cation. Zn+ 1s22s22p63s23p63d104s The shape of an orbital is given by the quantum number The atomic number of an element is 35. Atoms will gain or lose electrons to look like the nearest noble gas because the noble gases are unreactive due to the stability of having eight electrons in the highest energy level. It is willing to lose 2 electrons so that it has the same electron arrangements as the nearest noble gas, which is neon (2, 8). The mass number is an integer representation of the total number of protons and neutrons present in the isotope nucleus. These are atoms of the same element, so they have the same number of protons, but they have different numbers of neutrons. This corresponds to the \(2+\) charge formed when magnesium forms an ion. number of electrons beyond the previous element with a filled-shell electron configuration. Mass Number: The mass number of an atom is a concept used to describe isotopes of a chemical element. Note that when 3d block elements form ions, the 4s. The electron arrangement also shows the number of valence electrons which is two for magnesium because there are two electrons in the \(n=3\) energy level which is the highest occupied energy level for magnesium. diagram explaining why zinc ion complexes compounds are colourless of white no 3d electrons can be.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
